Biosensors and Chemical Biology Probes of Biomolecular Condensates
|
Biomolecular condensates present unique subcellular zones within cells that may have a chemical environment that diverges from the external cellular milieu. However, we are curious about the nature of this chemical environment and how it impacts the function of enzyme clients within these assemblies. To characterize this membraneless organelle environment, we're developing new biosensor and chemical biology approaches.
|
Design of a FRET Biosensor to Track Kinase Function in Condensates
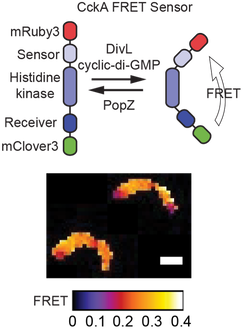
As discoveries of biomolecular condensates across all kingdoms of life rapidly emerge, we need new technologies to “visualize” how biomolecular condensates impact enzyme clients. Towards this goal, we engineered a biosensor to image how biomolecular condensates impact the structure of an essential signaling protein. The basis of this work was the histidine kinase CckA. This signaling protein localizes in two distinct biomolecular condensates with distinct client proteins that could alter CckA’s function. We incorporated donor and acceptor fluorescent proteins into the histidine kinase fold to construct the biosensor. Conformational domain re-arrangements within CckA alter the distance and orientation of donor and acceptor proteins leading to FRET changes. Our studies revealed that signals sensed by CckA alter FRET and that CckA exists in distinct structural states within the old and the new cell pole biomolecular condensates (see image to left). Thus, using this technology, we tested existing models in which the different activity states of CckA at the cell poles enable daughter cells to develop into distinct cell types upon division. Moreover, we showed that histidine kinase inhibitor binding to the FRET sensor could also be detected. This allows screening for new antibiotics that target histidine kinases (Duvall, 2020 ACS Sensor).